Lab 6d
Purpose
What plant materials, found locally, contain active ingredients that will inhibit the growth of bacteria?
Materials
|
-Balance, weigh boat, lab scoops
-LB broth base -Media bottles, 250 mL -Sterilizer/autoclave -Water bath, 37 C, shaking -Sterile LB agar -Lamar flow hood and disinfectant -Glasses, safety, plastic bunsen burner and gas lighter |
-Inoculating loop, Ni/Cr wire
-Petri Dishes, 60 X 15, sterile -E. Coli JM109 (stock plate) -Plant specimen -Mortar and pestle -Pipet, 10 mL and pump -Plastic Funnels, short stemmed -Filter paper disks, 5 mm diameter -Beakers, 100 mL |
-Syringe, 10 mL and filter, 0.2 um
-Reaction tubes and rack, 1.7 mL -Methanol, absolute -Pipet, 1 mL and pump -Dry block heater/heat block -Forceps, fine topped -Ampicillin -Glass spreader -Incubator oven, 37 C |
Procedure
Part II: Preparing Plant Extracts
4. Use a mortar and pestle to grind 2 g of plant leave or bark with 10 mL of deionized water. Then let it sit for 3 minutes. Filter the sample through an 11-cm filter paper funnel. Filter sterilize the filtered extract using a syringe filter. Collect 1 ml of extract into a 1.7 mL microtube. Label the sample.
5. Repeat step 4, but replace the water with methanol as the extracting solvent. After the methanol extraction, place the 1.7 mL tube with the 1 mL of methanol extract in a 65 C heat block (caps open) for 24 hours or more, if necessary, to evaporate the methanol. Reconstitute dry matter in the tube with 1 mL of deionized water.
7. Using sterile forceps (flamed in alcohol) drop three filter paper disks into each tube of filtered extract.
8. Prepare negative control disks, three each, of only methanol and only sterile distilled water.
9. Prepare 6 positive control disks of ampicillin solution.
10. Allow the disks sufficient time to soak up enough extract to be saturated (perhaps overnight)
11. Close the tubes. Store all samples at 4 C until ready to use.
12. Add 1 ml of sterile water to methanol extract tube and vortex
13. Using flame sterilized forceps, put 2 disks in each tube of extract
14. Using flame sterilized forceps put 2 discs in a microtube with water, positive control
15. Using sterile forceps put 1 disc in a microtube with amp, positive control
Part III: Setting Up Antimicrobial Plant Extract Assay
16. Transfer 1 mL of E. Coli to petri dish, which is labeled into 4 quadrants
17. Spread around with sterilized spreading loop
18. Let E. Coli soak into gel
19. Use sterile forceps to place disks in quadrants: 2 H2O extracts in two separate quadrants, 2 Methanol extracts in two separate quadrants, 1 amp in center for + control, one sterile H2O in center for -control
20. Incubate at 37 C for 24 hours
4. Use a mortar and pestle to grind 2 g of plant leave or bark with 10 mL of deionized water. Then let it sit for 3 minutes. Filter the sample through an 11-cm filter paper funnel. Filter sterilize the filtered extract using a syringe filter. Collect 1 ml of extract into a 1.7 mL microtube. Label the sample.
5. Repeat step 4, but replace the water with methanol as the extracting solvent. After the methanol extraction, place the 1.7 mL tube with the 1 mL of methanol extract in a 65 C heat block (caps open) for 24 hours or more, if necessary, to evaporate the methanol. Reconstitute dry matter in the tube with 1 mL of deionized water.
7. Using sterile forceps (flamed in alcohol) drop three filter paper disks into each tube of filtered extract.
8. Prepare negative control disks, three each, of only methanol and only sterile distilled water.
9. Prepare 6 positive control disks of ampicillin solution.
10. Allow the disks sufficient time to soak up enough extract to be saturated (perhaps overnight)
11. Close the tubes. Store all samples at 4 C until ready to use.
12. Add 1 ml of sterile water to methanol extract tube and vortex
13. Using flame sterilized forceps, put 2 disks in each tube of extract
14. Using flame sterilized forceps put 2 discs in a microtube with water, positive control
15. Using sterile forceps put 1 disc in a microtube with amp, positive control
Part III: Setting Up Antimicrobial Plant Extract Assay
16. Transfer 1 mL of E. Coli to petri dish, which is labeled into 4 quadrants
17. Spread around with sterilized spreading loop
18. Let E. Coli soak into gel
19. Use sterile forceps to place disks in quadrants: 2 H2O extracts in two separate quadrants, 2 Methanol extracts in two separate quadrants, 1 amp in center for + control, one sterile H2O in center for -control
20. Incubate at 37 C for 24 hours
Data/Analysis
Results:
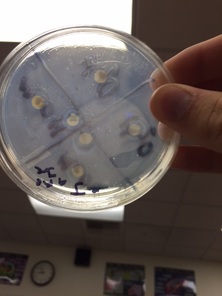
+ Control- This showed negative results, however our negative control was positive, so we must have switched them around.
- Control- This showed positive results because it was switched on accident.
MeOH Disc- Neither disc had large enough outlines to show they were positive. Both were about 0.5 cm diameter
H2O Disc- Neither disc had large enough outlines, outlines were also 0.5 cm for the water disc.
* we use methanol and water because water is polar and methanol is not. Therefore, non polar molecules in the plant can be extracted by the non polar methanol. Vice verse the polar molecules in the plant can be extracted by the polar water.
Analysis:
Did any extract give you positive results?
No, neither our water or methanol extracts gave positive results.
Did your controls work as expected?
Yes, but they were in the wrong locations. Our positive showed up negative and our negative showed up positive.
Discuss errors that could give you false results.
I noticed that grinding our plants leaves was very difficult and we hardly got any plant material grinned up. Therefore, we may not have had enough plant material to actually have the experiment work. Also, the disks were moved around a lot on the plates because it was our first time using them. That could have effected the growth of the bacteria in areas where we spread it out. Lastly, there could have been errors such as not sterilizing equipment properly or not waiting long enough for the disc to absorb.
Discuss further experimentation possibilities.
Next, we could use different types of bacteria to see which ones the plant resist. Also, we could use different solution than water and methanol to see what the plant molecules are attracted to. Lastly, we could figure out which molecules are in the plant and see how they are effecting the results.
Thinking like a bio technician:
1. If an extract gives a negative result in the antimicrobial assay, does that mean the extract is not an antimicrobial agent?
Yes, unless there is an error with the way we made the disks.
2. In preparing for the sample disks, some of the methanol extraction smell like alcohol. Why is that a problem?
Because you wouldn't know if the alcohol killed the bacteria or if the molecules did.
3. Each extract may have one or more compound in it. What should be done to begin to identify the exact compound in an extract that is causing the antimicrobial action?
To test each individual molecule, we would have to extract each compound. We could do this using polar and non polar chromatography. Then we would have to test each compound individually.
+ Control- This showed negative results, however our negative control was positive, so we must have switched them around.
- Control- This showed positive results because it was switched on accident.
MeOH Disc- Neither disc had large enough outlines to show they were positive. Both were about 0.5 cm diameter
H2O Disc- Neither disc had large enough outlines, outlines were also 0.5 cm for the water disc.
* we use methanol and water because water is polar and methanol is not. Therefore, non polar molecules in the plant can be extracted by the non polar methanol. Vice verse the polar molecules in the plant can be extracted by the polar water.
Analysis:
Did any extract give you positive results?
No, neither our water or methanol extracts gave positive results.
Did your controls work as expected?
Yes, but they were in the wrong locations. Our positive showed up negative and our negative showed up positive.
Discuss errors that could give you false results.
I noticed that grinding our plants leaves was very difficult and we hardly got any plant material grinned up. Therefore, we may not have had enough plant material to actually have the experiment work. Also, the disks were moved around a lot on the plates because it was our first time using them. That could have effected the growth of the bacteria in areas where we spread it out. Lastly, there could have been errors such as not sterilizing equipment properly or not waiting long enough for the disc to absorb.
Discuss further experimentation possibilities.
Next, we could use different types of bacteria to see which ones the plant resist. Also, we could use different solution than water and methanol to see what the plant molecules are attracted to. Lastly, we could figure out which molecules are in the plant and see how they are effecting the results.
Thinking like a bio technician:
1. If an extract gives a negative result in the antimicrobial assay, does that mean the extract is not an antimicrobial agent?
Yes, unless there is an error with the way we made the disks.
2. In preparing for the sample disks, some of the methanol extraction smell like alcohol. Why is that a problem?
Because you wouldn't know if the alcohol killed the bacteria or if the molecules did.
3. Each extract may have one or more compound in it. What should be done to begin to identify the exact compound in an extract that is causing the antimicrobial action?
To test each individual molecule, we would have to extract each compound. We could do this using polar and non polar chromatography. Then we would have to test each compound individually.